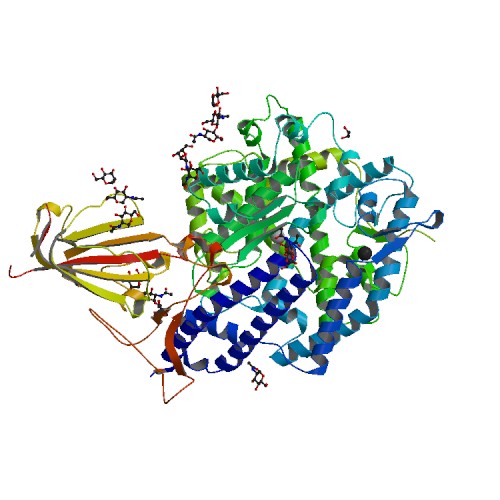
Structural basis of receptor recognition by SARS-CoV-2.
Publication Type:
Journal ArticleSource:
Nature (2020)Abstract:
<p>A novel SARS-like coronavirus (SARS-CoV-2) recently emerged and is rapidly spreading in humans. A key to tackling this epidemic is to understand the virus's receptor recognition mechanism, which regulates its infectivity, pathogenesis and host range. SARS-CoV-2 and SARS-CoV recognize the same receptor - human ACE2 (hACE2). Here we determined the crystal structure of the SARS-CoV-2 receptor-binding domain (RBD) (engineered to facilitate crystallization) in complex with hACE2. Compared with the SARS-CoV RBD, a hACE2-binding ridge in SARS-CoV-2 RBD takes a more compact conformation; moreover, several residue changes in SARS-CoV-2 RBD stabilize two virus-binding hotspots at the RBD/hACE2 interface. These structural features of SARS-CoV-2 RBD enhance its hACE2-binding affinity. Additionally, we show that RaTG13, a bat coronavirus closely related to SARS-CoV-2, also uses hACE2 as its receptor. The differences among SARS-CoV-2, SARS-CoV and RaTG13 in hACE2 recognition shed light on potential animal-to-human transmission of SARS-CoV-2. This study provides guidance for intervention strategies targeting receptor recognition by SARS-CoV-2.</p>